|
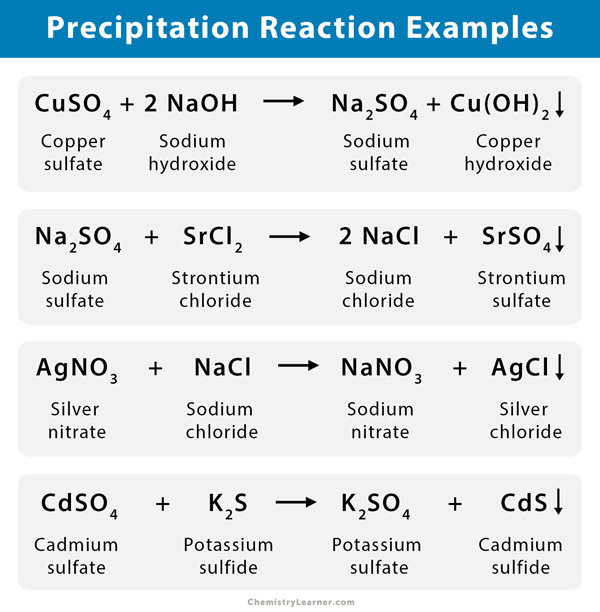
How do you know if a reaction is precipitating? Solubility depends on temperature, pressure, and polarity.ġ.To determine a precipitate, one must know the solubility rules.Precipitation reaction occurs when cations and anions in an aqueous solution combine to form an insoluble ionic solid termed precipitate.Carbonates, sulfides, and phosphates are insoluble.Most bromides, iodides, and chlorides are soluble exceptions are ones containing Ag⁺, Hg₂²⁺, Pb²⁺.Most hydroxides are considered insoluble, exceptions would be alkali metal hydroxides, and Ba(OH)₂.Ca(OH)₂ are slightly soluble.Nitrates, perchlorates, and chlorates are soluble.SOLUBILITY RULES AND PRECIPITATION REACTIONS An example would be sugar which is a polar solute, and it dissolves exceptionally well in polar water however, it remains insoluble in non-polar solvents like benzene. Polarity: A solute would dissolve best in a solvent with a similar chemical structure. 
This is very much visible when you open a soda bottle and experience a hissing sound which happens because the content has been under pressure to ensure the soda stays carbonated. Pressure: Pressure has very little effect on the solubility of solids and liquids but has a drastic effect on the gaseous solutes. However, with gasses, solubility decreases with an increase in temperature. For solids that dissolve in liquid, the solubility changes with changing temperature.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
